Contact information
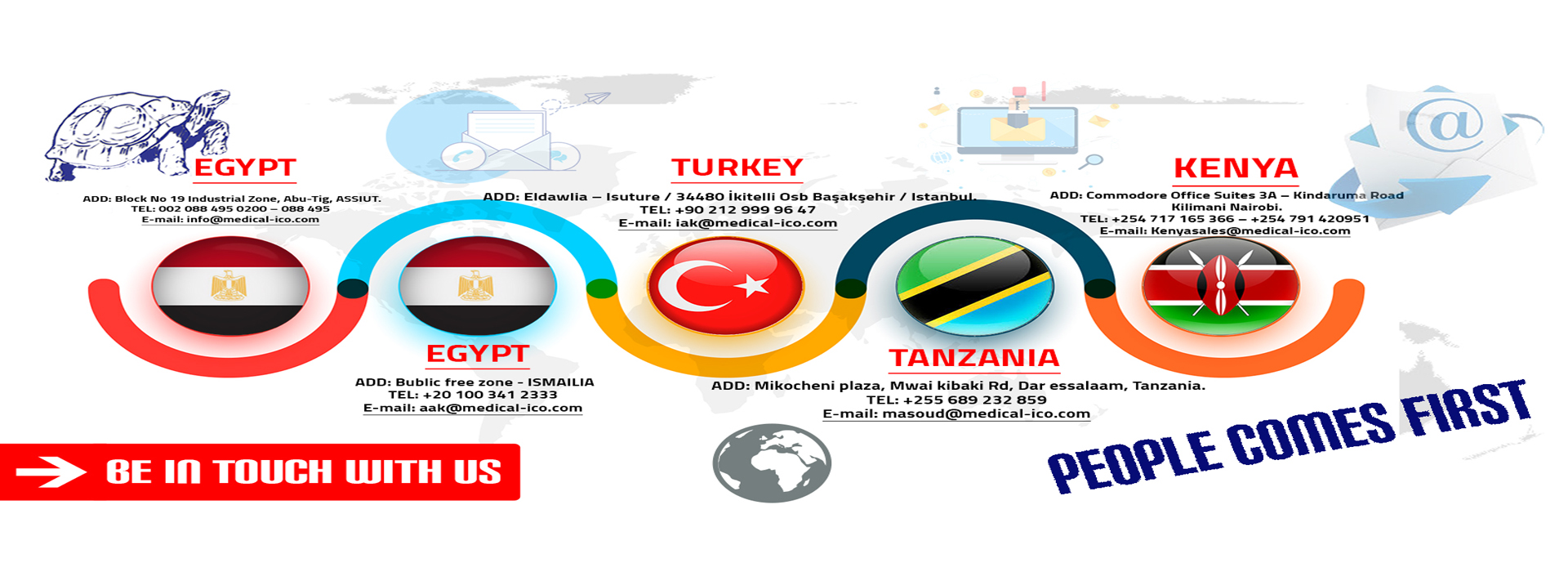
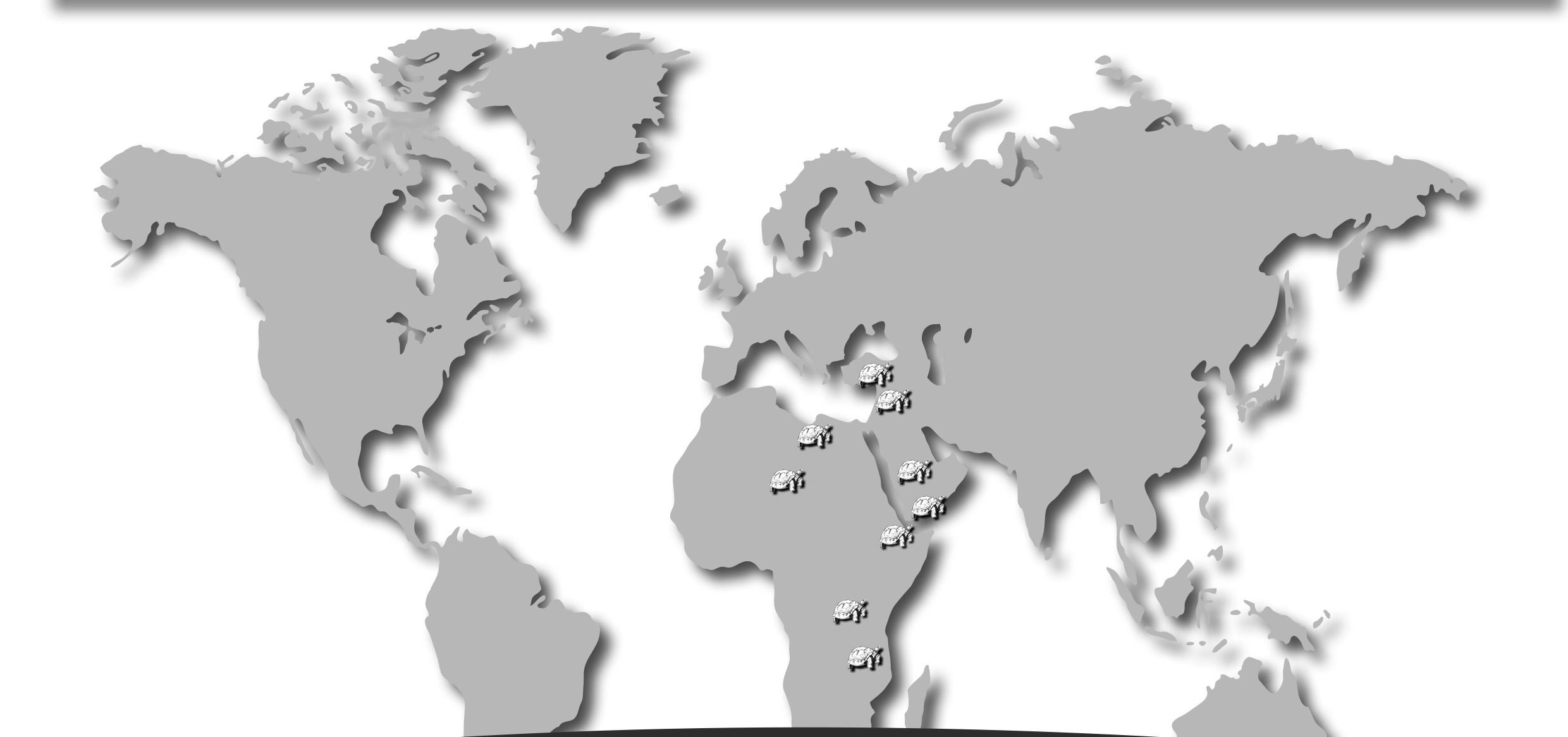
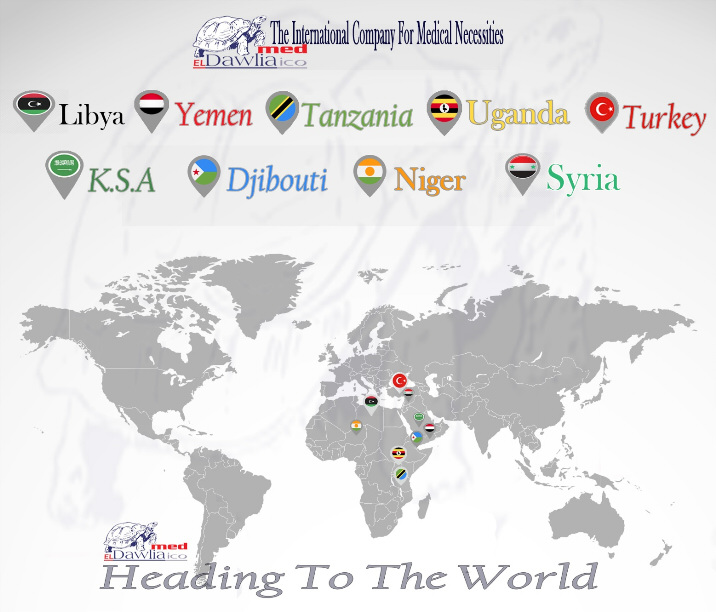
Egypt:
Asuit :
Address :Block No 19 Industrial Zone, Abu-Tig, Assuit-Egypt
Email:elsarry.e@medical-ico.com
Tel. :+201018001119
Ismaillia:
Adress: Ismailia Free Zone – Bublic free zone-Ismaillia-Egypt
Email: sales@medical-ico.com
Tel. :00201200505672
Turkey:
Adress: Eldawlia – Isuture -34480 İkitelli Osb Başakşehir – Istanbul-Turkey
Email:mgh@medical-ico.com
Tel:+90 212 999 96 47
Tanzania:
Adress:Mikocheni plaza, Mwai kibaki Rd, Dar essalaam, Tanzania
Email:iak@medical-ico.com
Tel:+255733333332
www.medical-ico.com